
At some point a couple of billion years ago, one cell tried to digest another and failed. The result was the first eukaryote, a complex cell type that today makes up all plants, animals and fungi: pretty much any organism you can see without a microscope. Eukaryotic cells are those that contain endosymbionts, the descendants of that original undigested cell. They are the mitochondria that power our cells, and the chloroplasts that allow plants to photosynthesize. Although they’ve been with us pretty much forever, they live separate lives, maintaining their own genomes, producing their own proteins, and exchanging them amongst each other. At least, we thought they did. But a new study from the University of Guelph has overturned a century of dogma by suggesting that chloroplasts may live as independently from one another as they do from the plant cells that host them.
It is well established that bacteria can exchange DNA and proteins between cells. “Bacteria and plastids actually share some of their outer wall components, so people thought that they might have retained this capability,” says Jaideep Mathur, Associate Professor in the Department of Molecular & Cellular Biology at the University of Guelph. (A plastid is a general term that includes chloroplasts and other chloroplast-like structures within plant cells; not all of them actively photosynthesize). We do know that plastids can create structures called stromules, tube-like extensions of the cell that can allow them to connect to each other. As long ago as 1888, a German microscopist named G. Haberlandt reported seeing what appeared to be two bulbous chloroplasts connected by a thin strand. “But that paper itself noted that if a single plastid is in the process of dividing, it can appear as two connected bodies,” says Mathur. “This is not the same as fusion between chloroplasts using stromules.”
It’s possible to use chemical dyes to distinguish plastids from one another, but those usually end up killing the cell. Evidence that chloroplasts actually exchange material would have to wait until the rise of the green fluorescent protein (GFP). Originally isolated from jellyfish, this biomolecule glows green when subjected to UV light. Its gene sequence was decoded in 1992, and since that time scientists have managed to get it expressed in everything from cats to fruit fly sperm, allowing them to visualize components of living cells. In 1997, a group from Cornell University took plant cells in which plastids expressed GFP and bleached them by subjecting them to high-intensity laser blasts. Single plastids remained bleached. However, if the laser bleached only one of a connected pair of plastids, the colour returned after a few seconds. This was taken as evidence that plastids can exchange proteins. But as Mathur points out, this could just as easily have been a single plastid undergoing division, rather than two separate plastids joining together. “If there is a connection, when was that connection established?” asks Mathur.
Over the last year, Mathur and his team have been working with a different protein, one originally isolated from brain coral. It too glows green, but when zapped with ultraviolet light, a molecular bond is broken that changes it irreversibly to red. This enabled them to finally distinguish between a plastid in the process of dividing and two separate plastids coming together. Experiments with mitochondria showed that when a green one and a red one meet, an intermediate orange colour is observed, proving that the two are exchanging proteins. But when chloroplasts and other plastids meet up with each other, the colours remain separate.
“We spent the whole of last year trying to uncover even one single event,” says Mathur. “We really dug around, and even used mutants which plastids 50 times larger than normal. It didn’t happen.” Mathur admits that it’s hard to prove a negative. “Who knows, tomorrow somebody could produce a convincing movie,” he says. “But we had thousands of cells with hundreds of plastids producing stromules all the time. If this is such a rare event that we cannot catch it in one year, how come the others caught it in a few minutes?” The paper in which Mathur’s group describe their findings is published online in The Plant Cell.
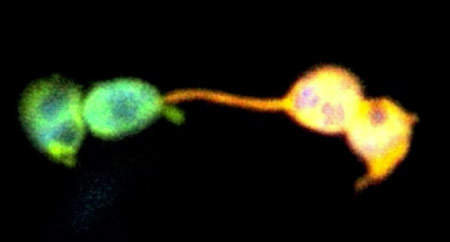
As happens so often in science, if Mathur’s theory is right, it only opens up more questions, the primary one being: if stromules aren’t used to exchange proteins, what are they for? It may be that they exchange other biomolecules than proteins (such as sugars or lipids) or are used for interplastid communication in a way that we still don’t understand. Any of these possibilities would open up new areas of research, about which Mathur is clearly excited. In fact, he believes that the separate lives of chloroplasts might be part of the explanation for why plants have been so successful in colonizing so many places on earth:
Animals and plants share the same environment. But if that environment changes a little, perhaps getting hotter or colder, we can run away, whereas plants stay in the same place. So the question is: is there something special that allows plants to withstand a lot of changes in their environment? The major difference between plant and animal cells is the plastid. A single cell in a leaf has about 120 of them. So imagine that instead of having a homogeneous population, the cell actually has 120 unique factories, each producing their own unique kind of starch or sugar or vitamin. To me, that variability is the fundamental reason why plants are able to survive.
1 Comment
[…] on tylerirving.ca Share this:TwitterFacebookLike this:LikeBe the first to like this post. « Functional […]